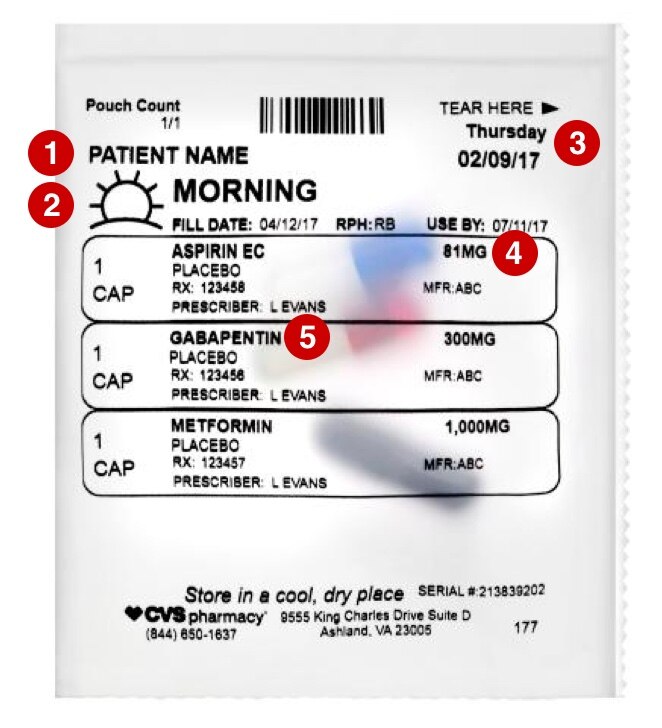
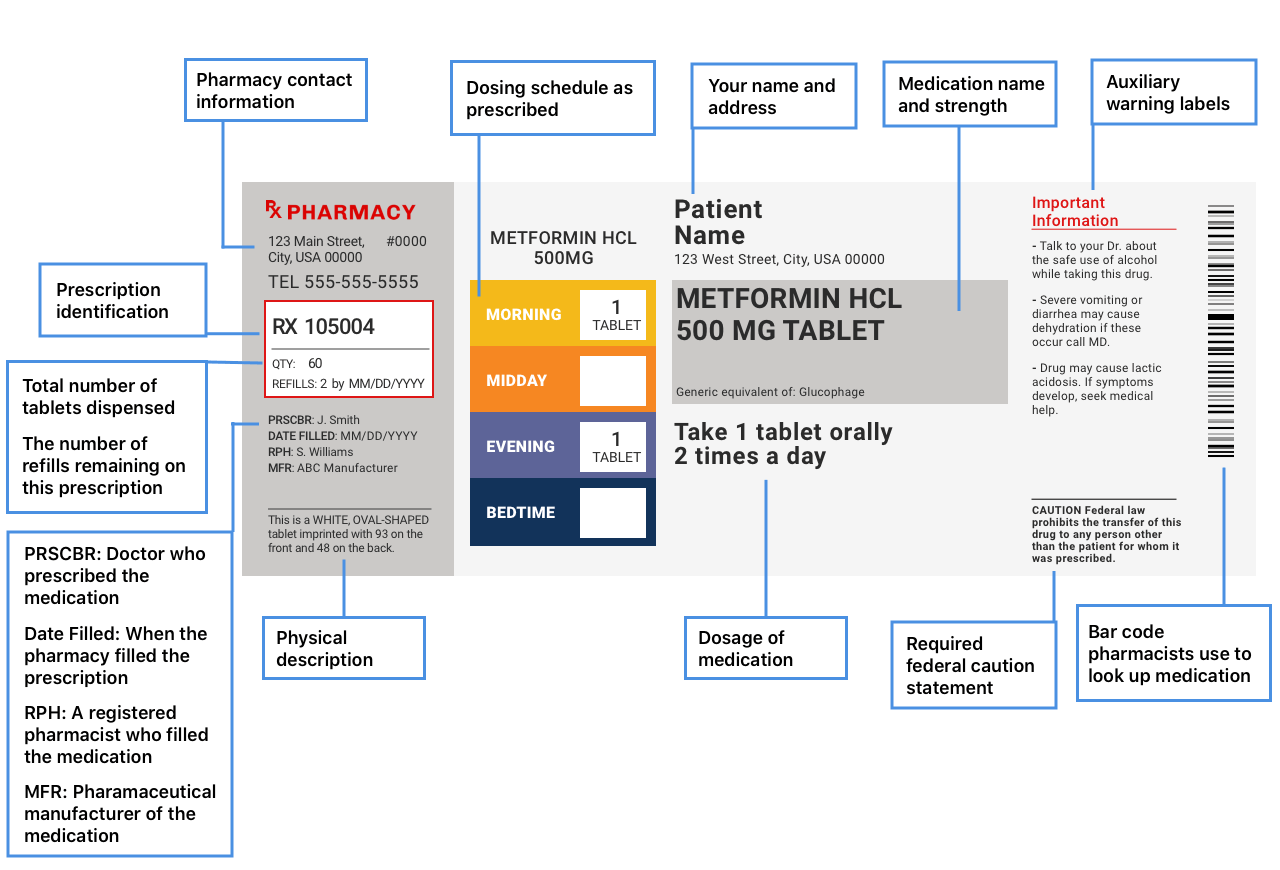
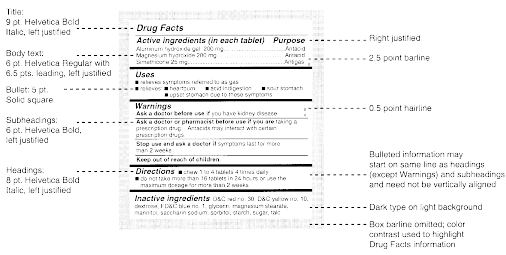
42 medication labels must include
Drug Labeling - StatPearls - NCBI Bookshelf Sep 24, 2022 ... Definition/Introduction · Highlights (a concise summary of label information) · Full prescribing Information · Limitations Statement · Product Names. What Information Should Be on Drug Labels? - MedicineNet Feb 3, 2022 ... Labeling requirements for prescription drugs · Statement of identity · Brand name · Net quantity of contents · Statement of dosage.
CFR - Code of Federal Regulations Title 21 - FDA (1) The labeling must contain a summary of the essential scientific information needed for the safe and effective use of the drug. (2) The labeling must be ...

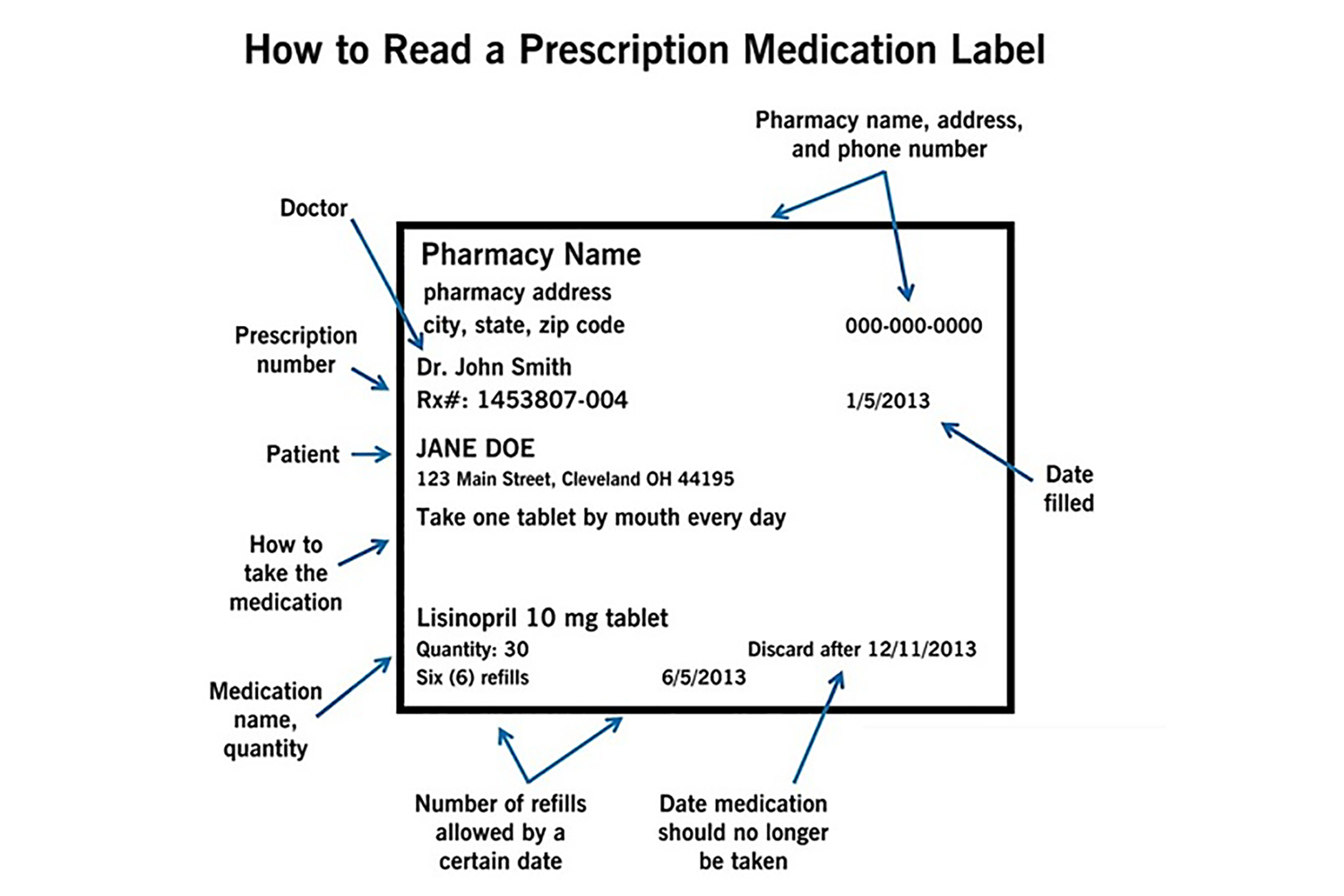
Medication labels must include
How to Label a Medical Syringe Mar 14, 2022 ... Unit dose medication packaging solutions including materials like labels for single-dose vials and syringes must also comply with federal ... Drug labelling - Wikipedia Drug labelling is also referred to as prescription labelling, is a written, printed or graphic matter upon any drugs or any of its container, ... Understanding the Critical Requirements for FDA Drug Labels FDA regulations require that all medication labels include: · Name of Product · Table of Drug Facts · Active Ingredients · Proper Use and Purpose · Warnings ...
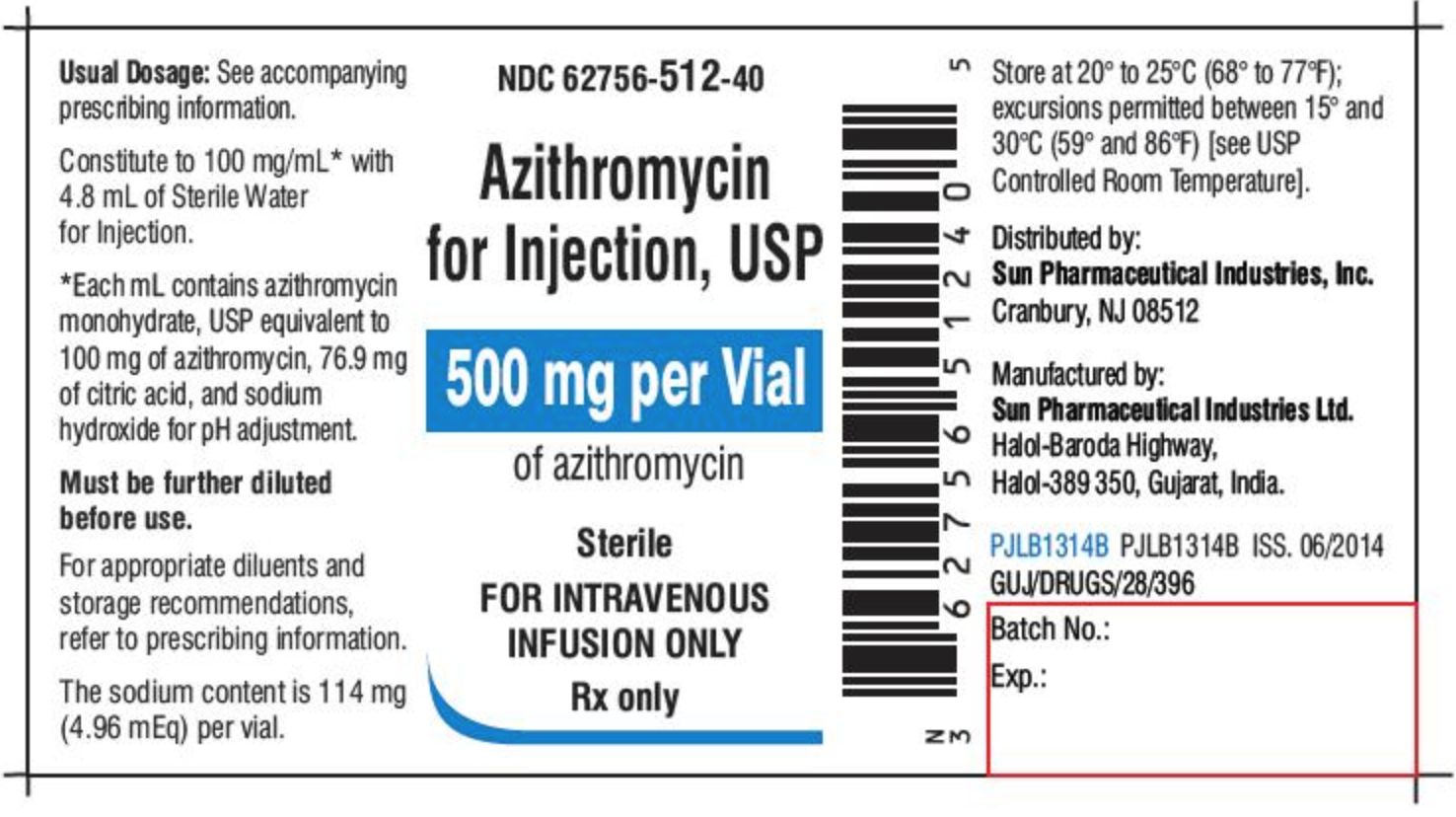
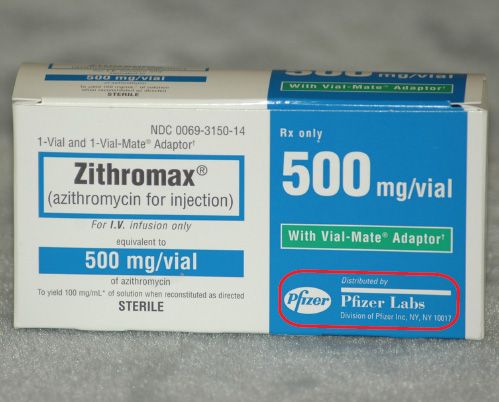
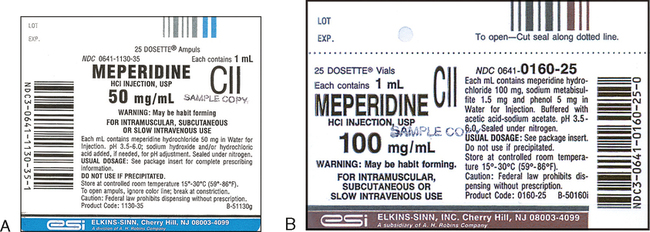
Medication labels must include. Medication Labels 101: Categories, Regulations, and Best Practices The basics of medication labels include the drug name, dosage, and directions. Medication labels should always include warnings regarding safety. How to read prescription drug labels - BeMedwise All prescription medicine containers include information on the label including the patient's name, the name of the medicine, dosage and instructions on how ... FDA Issues New RX Label Requirements - The Recovery Village Apr 20, 2022 ... A human prescription drug label must include all of the legal information set forth by the FDA. Details on Abuse-Deterrent: The information that ... Statement on Labeling of Pharmaceuticals for Use in Anesthesiology Label content: Syringes: The drug's generic name and concentration (in units per mL) should be the most prominent items displayed on the label of each syringe.
Understanding the Critical Requirements for FDA Drug Labels FDA regulations require that all medication labels include: · Name of Product · Table of Drug Facts · Active Ingredients · Proper Use and Purpose · Warnings ... Drug labelling - Wikipedia Drug labelling is also referred to as prescription labelling, is a written, printed or graphic matter upon any drugs or any of its container, ... How to Label a Medical Syringe Mar 14, 2022 ... Unit dose medication packaging solutions including materials like labels for single-dose vials and syringes must also comply with federal ...




















This comment has been removed by the author.
ReplyDeletePharmaceutical quality assurance companies often work closely with pharmaceutical manufacturers to implement best practices in labeling. This involves staying abreast of industry developments, incorporating the latest technologies, and ensuring that labeling processes align with the highest standards.
ReplyDelete